My mother died of Alzheimer’s at the age of 69, so I can personally attest to the horror of this disease. I can think of few things worse than slowly watching your cognitive abilities decline, particularly if you are aware of the progressive deterioration as my mother was. So I’m keeping a close watch on the latest Alzheimer’s research, including the research of my colleague William Jagust who is a neuroscientist at UC Berkeley.
Dr. Jagust is participating in the Alzheimer’s Disease Neuroimaging Initiative (ADNI), which is large multicenter project supported by NIH, private pharmaceutical companies and nonprofit organizations. The primary goal of ADNI is to discover indicators (biomarkers) that can track disease progression and hopefully diagnose Alzheimer’s early on. Basically, they want to help speed up and streamline drug and clinical trials by developing biomarkers that track Alzheimer’s more reliably.
The initial ADNI five-year research project completed last fall. It studied cognition, function, brain structure and biomarkers for 800 subjects (200 elderly controls, 400 subjects with mild cognitive impairment, and 200 subjects with Alzheimer’s). The clinical data from the patients went into a large database, including MRI scans, PET scans, blood tests, neuropsychological tests, and genetic tests. The truly unique thing is that this database can be accessed by the public through a website. Basically the raw data (with patient personal information removed) is made available for everyone to use, in hopes that this will help scientists more rapidly understand and treat Alzheimer’s. This ADNI project just received the second phase of funding, so the studies will be expanded.
Although the cause and progression of Alzheimer’s disease is not fully understood, current research indicates that the disease is associated with the formation of “amyloid plaques” and “neurofibrillary tangles” in the brain that damage nerve cells. What does this mean? Amyloid plaques are protein fragments that the body produces naturally. In a healthy brain, these protein fragments are broken down and eliminated. In a brain with Alzheimer’s, the fragments instead accumulate to form hard, insoluble plaques between nerve cells. This excess amyloid buildup occurs before clinical Alzheimer’s symptoms, so it may be used as a predictor of disease. Neurofibrillary tangles are insoluble twisted fibers found inside the brain’s cells. These tangles mainly consist of a protein called tau, which helps form microtubules that transport nutrients from one part of the nerve cell to another. In an Alzheimer’s brain, the tau protein is abnormal and the tangles collapse this important transport system.
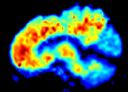
Dr. Jagust and other researchers are studying this beta-amyloid buildup using medical imaging, including PET imaging with a new drug called [11C]Pittsburg Compound B. This new PET drug binds to beta-amyloid plaques and indicates their size and position. “With PET, we’re able to study the biochemistry of the brain, and with MRI we can study both the anatomy and structure of the brain,” Jagust said. “We can also study some of the function of the brain to see what parts of the brain are active during different cognitive tests. So when you put all this information together, you can get a very detailed picture of how the brain is functioning and how function and structure might change with age.” Last fall Jagust published an article on the relationships between biomarkers in aging and dementia. The group found that the confluence of three factors — beta-amyloid deposition, atrophy of the hippocampus (part of the brain that stores and sorts memories), and episodic memory loss — signals early stage of Alzheimer’s. Hopefully this new understanding will ultimately provide early and more accurate diagnosis.
I don’t have room here to summarize all the results from the Jagust lab, let alone all the other labs doing Alzheimer’s research. But I must say that I’m optimistic given the recent progress they have made in understanding the disease. There are also many clinical trials underway for new Alzheimer’s drugs, including ones that hope to stop cognitive deterioration instead of just reducing symptoms. I’m encouraged but I still tell my friends who are doing the research that they need to find a cure within the next 10 years, because I do not want to suffer through this frightening disease like my mother did.
